Biobeat Gets FDA Clearance for Wearable Blood Pressure Monitors

Biobeat Gets FDA Clearance for Wearable Blood Pressure Monitors
Biobeat received FDA 501K clearance for its patch and smartwatch for measurement of blood pressure, oxygenation and heart rate.

Biobeat ha hecho una publicación en LinkedIn

7 Eye-Catching Wearables at MWC19

BioBeat - Products, Competitors, Financials, Employees, Headquarters Locations

Actiste's CE marks, launch of Care, Biobeat patch FDA clearance
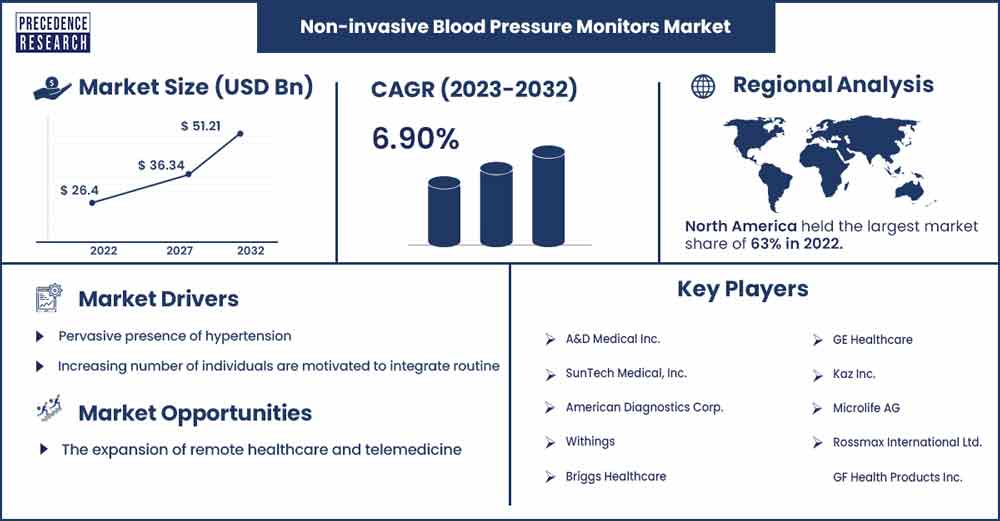
Non-invasive Blood Pressure Monitors Market Size To Rise USD 51.21 Bn By 2032

Wearable Health Monitors: Do They Work? - WSJ

Aktiia to bring continuous blood pressure monitor to US

Portfolio – Infinity Medical

Biobeat gets CE mark for AI-based remote patient monitoring platform

Hypertension: Connected Device Applications and Opportunities, by Sequenex

BioBeat Chest Patches and COVID-19 - Incarta


